
Unexpectedly, the cell presented a short circuit after almost 900 charge/discharge cycles, which is similar to that using a Li metal anode. In the present work, we cycled a full cell at high current (3.8 mA cm −2) and high loading (4 mAh cm −2) to investigate the interface stability between the sulfide electrolyte and Li-In anode. There have been few investigations to clarify the issue. 
However, most batteries were cycled at low current (< 1 mA cm −2) and loading (< 1 mAh cm −2), and it is unclear whether the Li-In alloy anode is still stable toward SSEs at increased current, which is critical for high-power applications of ASSLBs. Li-In alloys exhibit favorable long-cycling stability toward SSEs in ASSLBs 25, 26, 27. Li-In alloys are usually considered thermodynamically and kinetically stable materials toward SSEs and are widely used in the laboratory for testing the performance of electrolytes or cathodes. Li +/Li) over a wide stoichiometry range 15. In addition, the insertion of lithium into other metals can decrease the lithium chemical potential and suppress the electrochemical decomposition of solid sulfide electrolytes (SSEs) 23, 24.Īmong various lithium alloys, Li-In alloys are particularly popular due to their mechanical ductility and constant redox potential (0.62 V vs. Generally, the alloy layer or bulk has a higher lithium diffusivity than pure lithium, which is favorable for lithium transport toward the interface thus, uniform lithium plating can be achieved 21, 22. A number of alloys including Li-Al 13, Li-In 14, 15, Li-Si 16, Li-Au 17, 18, 19, and Li-Sn 20 have been reported as interlayers or solid solutions for sulfide-based ASSLBs. Lithium alloys can be easily prepared by the solid-state diffusion method at ambient temperature. Lithium alloys provide an attractive alternative to construct a stable electrolyte-electrode interface that enables long-term cycling for ASSLBs 12. 
However, it is difficult for pure Li to be applied in sulfide-based ASSLBs in the short term because of the severe interfacial side reactions 6, 7, 8 and the growth of Li dendrites 9, 10, 11. the standard hydrogen electrode) and high theoretical specific capacity (3860 mAh g −1). Lithium metal is recognized as the most attractive choice for anode materials to achieve high energy density due to its low electrochemical potential (−3.04 V vs. 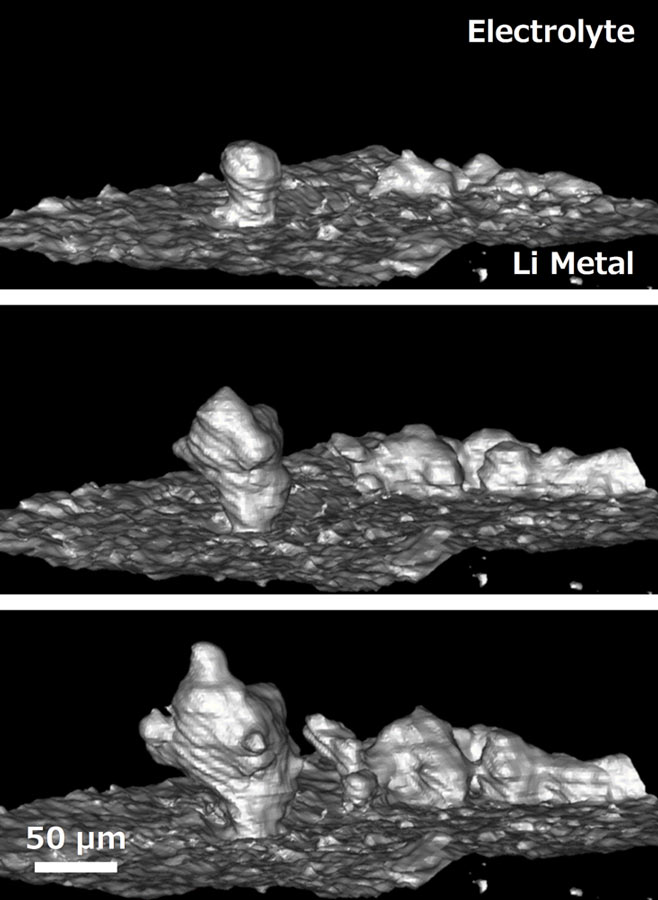
All-solid-state lithium batteries (ASSLBs) using nonflammable solid electrolytes may not only overcome safety concerns in LIBs but also achieve high energy density 1, 2, 3, 4, 5. The thermal instability of conventional lithium-ion batteries (LIBs), which originates from the intrinsic characteristics of liquid electrolytes, causes safety issues and has become a serious impediment to automotive applications. Furthermore, the difference between Li and Li-In dendrites is investigated and discussed to demonstrate the distinct type of dendrite morphology. Via ex situ measurements and simulations, we demonstrate that the irregular growth of Li-In dendrites leads to cell short circuits after room-temperature long-term cycling. In this work, we report the growth of Li-In dendritic structures when the alloy material is used in combination with a Li 6PS 5Cl solid electrolyte and Li(Ni 0.6Co 0.2Mn 0.2)O 2 positive electrode active material and cycled at high currents (e.g., 3.8 mA cm −2) and high cathode loading (e.g., 4 mAh cm −2). In this context, alloy materials such as lithium-indium (Li-In) alloys are widely used at the laboratory scale because of their (electro)chemical stability, although no in-depth investigations on their morphological stability have been reported yet. However, the application of lithium metal is hindered by issues associated with the growth of mossy and dendritic Li morphologies upon prolonged cell cycling and undesired reactions at the electrode/solid electrolyte interface. All-solid-state lithium-based batteries with inorganic solid electrolytes are considered a viable option for electrochemical energy storage applications.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
